A) K > Q
B) K < Q
C) K = Q
D) K = 1
E) Q = 1
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The formation constant for the reaction Ag+(aq) + 2NH3(aq)  Ag(NH3) 2+(aq)
Is Kf = 1.7 × 107 at 25°C. What is ΔG° at this temperature?
Ag(NH3) 2+(aq)
Is Kf = 1.7 × 107 at 25°C. What is ΔG° at this temperature?
A) -1.5 kJ
B) -3.5 kJ
C) -18 kJ
D) -23 kJ
E) -41 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔG° for the reaction of ammonia with fluorine. 2NH3(g) + 5F2(g) → N2F4(g) + 6HF(g) Substance: NH3(g) F2(g) N2F4(g) HF(g) ΔG°f (kJ/mol) : -16.4 0 9.9 -275.4
A) 179.1 kJ
B) -179.1 kJ
C) 1539.7 kJ
D) -1539.7 kJ
E) None of these choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the reaction 2Cl2(g) + SO2(g) → SOCl2(g) + Cl2O(g) Substance: Cl2(g) SO2(g) SOCl2(g) Cl2O(g) S°(J/K·mol) : 223.0 248.1 309.77 266.1
A) -118.2 J/K
B) -104.8 J/K
C) 104.8 J/K
D) 118.2 J/K
E) 1270.0 J/K
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second law of thermodynamics tells us that
A) the entropy of the universe is constant.
B) entropy is neither created nor destroyed.
C) the universe proceeds toward a state of lower entropy.
D) the universe proceeds toward a state of higher entropy.
E) the universe cannot create entropy.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the free energy change, ΔG°, for the equilibrium between hydrogen iodide, hydrogen, and iodine at 453°C? Kc = 0.020 2HI(g)  H2(g) + I2(g)
H2(g) + I2(g)
A) 6.4 kJ
B) 8.8 kJ
C) 15 kJ
D) 19 kJ
E) 24 kJ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the reaction 4Cr(s) + 3O2(g) → 2Cr2O3(s) Substance: Cr(s) O2(g) Cr2O3(s) S°(J/K·mol) : 23.77 205.138 81.2
A) -548.1 J/K
B) -147.7 J/K
C) 147.7 J/K
D) 310.1 J/K
E) 548.1 J/K
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following pairs will the first system have a higher entropy than the second? Assume P and T are the same for each pair, unless stated otherwise.
A) 1 mole He(g) ; 1 mole Kr(g)
B) 1 mole O2(g) ; 2 mole O(g)
C) 1 mole CH4(g) ; 1 mole C2H6(g)
D) 1 mole Xe(g) at 1 atmosphere; 1 mole Xe(g) at 0.5 atmosphere
E) 20 one-dollar bills distributed randomly among 20 people; 20 one-dollar bills distributed randomly among 10 people
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate ΔS° for the combustion of propane. C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) Substance: C3H8(g) O2(g) CO2(g) H2O(g) S°(J/K·mol) : 269.9 205.138 213.74 18.825
A) -100.9 J/K
B) -72.5 J/K
C) 72.5 J/K
D) 100.9 J/K
E) 877.5 J/K
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
"A diamond is forever" is one of the most successful advertising slogans of all time. But is it true? For the reaction shown below, calculate the standard free energy change at 298K and determine whether or not a diamond is "forever". C(diamond) → C(graphite) Data: ΔHf°(diamond) = 1.895 kJ/mol; S°(diamond) = 2.337 J mol-1K-1; S°(graphite) = 5.740 J mol-1K-1.
A) ΔG° = 2.19 kJ; forever
B) ΔG° = -1.90 kJ; not forever
C) ΔG° = -2.90 kJ; not forever
D) ΔG° = 1.90 kJ; forever
E) ΔG° = < -1000 kJ; not forever
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given: H2O(l) → H2O(g) ΔH° = 40.7 kJ at 373K What is the entropy change in the system (ΔS) when one mole of water vaporizes at 100°C and a pressure of one atmosphere?
A) 407 J/K
B) -407 J/K
C) 109 J/K
D) -109 J/K
E) J/K
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a spontaneous process, the entropy of the system always increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the figure that shows ΔG° for a chemical process plotted against absolute temperature. From this plot, it is reasonable to conclude that 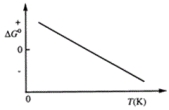
A) ΔH° > 0, ΔS° > 0
B) ΔH° > 0, ΔS° < 0
C) ΔH° < 0, ΔS° > 0
D) ΔH° < 0, ΔS° < 0
E) None of these choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is true for pure oxygen gas, O2(g) at 25°C?
A) ΔH°f > 0
B) ΔH°f < 0
C) ΔG°f > 0
D) ΔG°f < 0
E) S° > 0
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
In some spontaneous processes, the entropy of the surroundings decreases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of water is heated at a constant pressure of one atmosphere. Initially, the sample is ice at 260 K, and at the end the sample consists of steam at 400 K. In which of the following 5K temperature intervals would there be the greatest increase in the entropy of the sample?
A) from 260 K to 265 K
B) from 275 K to 280 K
C) from 360 K to 365 K
D) 370 K to 375 K
E) from 395 K to 400 K
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
For a given reaction, a change in the pressure may result in a change in the sign of ΔG.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a process with ΔS < 0, which one of the following statements is correct?
A) The process will definitely be spontaneous if ΔH < 0.
B) The process will be definitely be spontaneous if ΔH < TΔS.
C) The process can never be spontaneous.
D) The process will definitely be spontaneous, regardless of ΔH.
E) The process will definitely be spontaneous if ΔSsurr > 0.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which relationship or statement best describes ΔS° for the following reaction? O3(g) + NO(g) → O2(g) + NO2(g)
A) ΔS° ≈ 0
B) ΔS° < 0
C) ΔS° > 0
D) ΔS° = ΔH°/T
E) More information is needed to make a reasonable prediction.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For a reaction at equilibrium, ΔSuniv = 0.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 84
Related Exams