A) magnesium difluoride
B) magnesium fluoride
C) magnesium(II) fluoride
D) monomagnesium difluoride
E) None of these choices are correct
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the element bromine, the symbol, group number, group name and physical state are, respectively
A) Bo, 17, noble gas, gas.
B) B, 13, semimetal, solid.
C) Br, 17, halogen, liquid.
D) Br, 17, halogen, gas.
E) Br, 15, chalcogenide, gas.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements are the least reactive?
A) alkali metals
B) noble gases
C) halogens
D) alkaline earth metals
E) metalloids
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets are isoelectronic (i.e., have the same number of electrons) ? i. Br-, Kr, Sr2+ Ii. C, N-, O2- Iii. Mg2+, Ca2+, Sr2+ Iv. O2-, O, O2+ V. Ag+, Cd2+, Pd
A) i and ii
B) i and v
C) i, iii and iv
D) ii, iii and v
E) i, iii iv and v
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Tetrasulfur dinitride decomposes explosively when heated. What is its formula?
A) S2N4
B) S4N2
C) 4SN2
D) S4N
E) S2N
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barium fluoride is used in embalming and in glass manufacturing. Which of the following gives the formula and bonding for barium fluoride?
A) BaF2, ionic compound
B) BaF2, covalent compound
C) BaF, ionic compound
D) BaF, covalent compound
E) Ba2F, ionic compound
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the chemical symbol for the group 6A (16) element that lies in period 4?
A) Cr
B) Hf
C) W
D) Ti
E) Se
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The formula C9H20 is an empirical formula.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the modern periodic table, the order in which the elements are placed is based on
A) atomic mass.
B) mass number.
C) atomic number.
D) atomic size.
E) chemical reactivity.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a metal?
A) nitrogen, N, Z = 7
B) phosphorus, P, Z = 15
C) arsenic, Z = 33
D) thallium, Tl, Z = 81
E) silicon, Si, Z = 14
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is covalent?
A) CaCl2
B) MgO
C) Al2O3
D) Cs2S
E) PCl3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromine has two naturally-occurring isotopes. 79Br has a mass of 78.9 amu and accounts for 50.3% of bromine atoms. If the atomic mass of bromine is 79.9 amu, what is the mass of an atom of the second bromine isotope?
A) 77.9 amu
B) 80.0 amu
C) 80.1 amu
D) 80.9 amu
E) 88.9 amu
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
After an atom has lost an electron it becomes a/an ______ and has a _______ charge.
A) anion, positive
B) isotope, negative
C) anion, negative
D) cation, positive
E) nucleus, positive
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following symbols does not represent an element?
A) O2
B) Co
C) HF
D) Cs
E) Xe
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Sodium peroxide is an oxidizer used to bleach animal and vegetable fibers. What is its formula?
A) NaO
B) NaO2
C) Na2O2
D) Na2O
E) NaH2O2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following formulas of ionic compounds is the least likely to be correct?
A) CaCl2
B) NaSO4
C) MgCO3
D) KF
E) Cu(NO3) 2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
J. J. Thomson studied cathode ray particles (electrons) and was able to measure the mass/charge ratio. His results showed that
A) the mass/charge ratio varied with as the cathode material was changed.
B) the charge was always a whole-number multiple of some minimum charge.
C) matter included particles much smaller than the atom.
D) atoms contained dense areas of positive charge.
E) atoms are largely empty space.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A column of the periodic table is called a
A) group.
B) period.
C) isotopic mixture.
D) pillar.
E) shell.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Bromine is the only nonmetal that is a liquid at room temperature. Consider the isotope bromine-81, 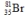 . Select the combination which lists the correct atomic number, neutron number, and mass number, respectively.
. Select the combination which lists the correct atomic number, neutron number, and mass number, respectively.
A) 35, 46, 81
B) 35, 81, 46
C) 81, 46, 35
D) 46, 81, 35
E) 35, 81, 116
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the formula for lead (II) oxide?
A) PbO
B) PbO2
C) Pb2O
D) PbO4
E) Pb2O3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 100
Related Exams