B) False
Correct Answer

verified
Correct Answer
verified
True/False
The more C  O and O
O and O  H bonds there are in a substance, the greater will be the amount of heat released when a fixed mass of the substance is burned.
H bonds there are in a substance, the greater will be the amount of heat released when a fixed mass of the substance is burned.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The electrostatic energy of two charged particles is inversely proportional to the distance between them.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) S
B) Ru
C) Si
D) Te
E) Cs
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the most polar bond amongst the following.
A) C ![]() O
O
B) Si ![]() F
F
C) Cl ![]() F
F
D) C ![]() F
F
E) C ![]() I
I
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the element whose Lewis symbol is correct.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
A hypothetical ionic substance will not form merely because it has a high lattice energy. Explain why, using energy-based arguments.
Correct Answer

verified
In order for an ionic substance to form,...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
As a measure of the strength of metallic bonding, the boiling point of a metal is a better indicator than its melting point.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Using appropriate, real examples to illustrate your answer, describe the correlation between bond energy and bond length for a series of varying bond order.
Correct Answer

verified
Carbon and oxygen form single, double an...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Describe, with appropriate explanations, the key factors which affect the magnitude of the lattice energy of an ionic substance.
Correct Answer

verified
By Coulomb's law, the energy of two elec...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Analysis of an unknown substance showed that it has a moderate melting point and is a good conductor of heat and electricity in the solid phase. Which of the following substances would have those characteristics?
A) NaCl
B) Si
C) CCl4
D) I2
E) Ga
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the compound with the highest (i.e., most negative) lattice energy.
A) CaS(s)
B) BaO(s)
C) NaI(s)
D) LiBr(s)
E) MgO(s)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following properties is least characteristic of substances composed of small, covalently-bonded molecules?
A) low melting point
B) low boiling point
C) weak bonds
D) poor electrical conductor when solid
E) poor electrical conductor when molten
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The melting points of metals are only moderately high because
A) metallic bonding is weak.
B) metals have fewer bonding electrons than non-metals.
C) metals also have relatively low boiling points.
D) the melting process does not break the metallic bonds.
E) metals prefer to be bonded to non-metals.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the least electronegative?
A) Si
B) Se
C) S
D) Sc
E) Sr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains covalent bonds?
A) BaO
B) IBr
C) Mg
D) LiBr
E) Cu
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Covalently bonded substances do not necessarily exist as separate molecules.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The lattice energy of MgCl2 is the energy change for which one of the following processes?
A) Mg(s) + Cl2(g) MgCl2(s)
B) Mg(g) + 2Cl(g) MgCl2(s)
C) Mg2+(s) + 2Cl¯(g) MgCl2(g)
D) Mg2+(g) + 2Cl¯(g) MgCl2(s)
E) MgCl2(aq) MgCl2(s)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When two atoms form a covalently-bonded diatomic molecule, the distance between the nuclei at which the potential energy is at a minimum is called
A) the bond energy.
B) the bond length.
C) the molecular diameter.
D) the covalent radius.
E) the covalent diameter.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The lattice energy of rubidium chloride is the energy change accompanying the process
Rb+(g) + Cl-(g) RbCl(s)
Calculate the lattice energy of RbCl using the following data: 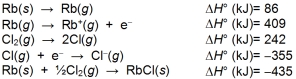
Correct Answer

verified
Lattice en...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 41 - 60 of 73
Related Exams