A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated bond is:
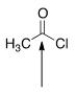
A) Nucleophilic because it is electron-deficient.
B) Electrophilic because it is electron-rich.
C) Nucleophilic because it is electron-rich.
D) Electrophilic because it is electron-deficient.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What intermolecular force is generally considered the weakest?
A) Hydrogen bonding
B) London dispersion forces
C) Dipole-dipole
D) Ion-ion
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the lowest boiling point?
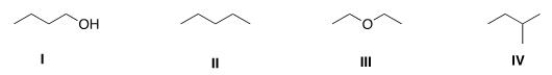
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following could most likely serve as an ionophore?
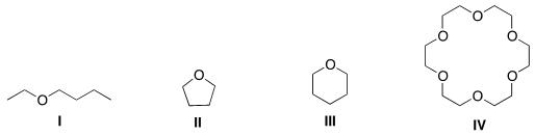
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a tertiary amine?

A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain the same functional groups?

A) I, II, III
B) I, II, IV
C) II, III, IV
D) I, III, IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point?

A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following lists contains common heteroatoms found in organic molecules?
A) N, O, S, P, Cl
B) Na, O, S, P, Cl
C) Na, Mg, S, N, Cl
D) Na, Mg, O, N, Cl
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds would be expected to be more soluble in hexane (C6H14) ?
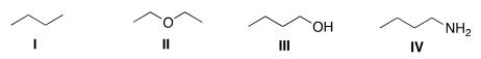
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of increasing melting point, putting the compound with the least melting point first. 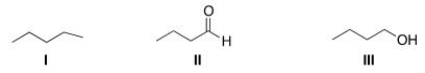
A) II < I < III
B) I < III < II
C) I < II < III
D) III < II < I
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following structures contains an alkene?

A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules can bond hydrogen to another molecule of itself?
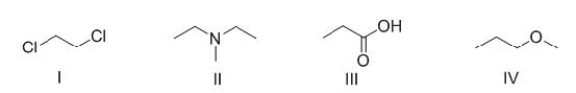
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The indicated carbon atom is:
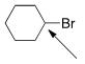
A) Electrophilic because it is electron-deficient.
B) Nucleophilic because it is electron-deficient.
C) Electrophilic because it is electron-rich.
D) Nucleophilic because it is electron-rich.
F) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following statements about vitamin A, drawn below, is true?
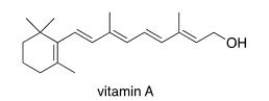
A) Vitamin A is soluble in H2O.
B) Vitamin A is insoluble in organic solvents.
C) Vitamin A contains an aromatic ring.
D) Vitamin A is insoluble in H2O.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly matches the molecules to the names of the functional group?
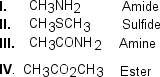
A) I and II
B) II and IV
C) III and IV
D) II and III
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following compounds in order of decreasing boiling point, putting the compound with the highest boiling point first. 
A) I > II > III > IV
B) III > IV > II > I
C) III > II > IV > I
D) I > IV > II > III
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements best describes the relationship between the surface area of a molecule and the strength of the intermolecular forces?
A) The larger the surface area, the weaker the intermolecular forces.
B) The larger the surface area, the stronger the intermolecular forces.
C) The smaller the surface area, the stronger the intermolecular forces.
D) There is no relationship between surface area and intermolecular forces.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the molecule donepezil (used to treat Alzheimer's disease) .Which of the following lists the correct functional groups present in donepezil?
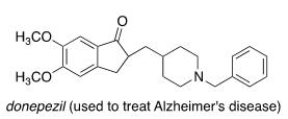
A) Amide, aromatic, ether, ketone
B) Amide, aromatic, ester, ketone
C) Amine, aromatic, ester, ketone
D) Amine, aromatic, ether, ketone
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Which of the following compounds has the highest boiling point?
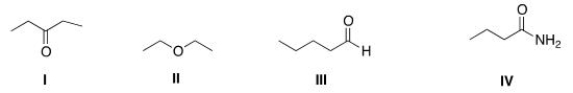
A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
D
Correct Answer
verified
Showing 1 - 20 of 45
Related Exams