A) 12.3 g
B) 24.7 g
C) 37.1 g
D) 49.4 g
E) 74.2 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Nitrogen will behave most like an ideal gas
A) at high temperature and high pressure.
B) at high temperature and low pressure.
C) at low temperature and high pressure.
D) at low temperature and low pressure.
E) at intermediate (moderate) temperature and pressure.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At what temperature in kelvin is the root mean square speed of helium atoms (atomic weight = 4.00) equal to that of oxygen molecules (molecular weight = 32.00) at 300. K?
A) 37.5 K
B) 75 K
C) 106 K
D) 292 K
E) 2400. K
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Freon-12, CF2Cl2, which has been widely used in air conditioning systems, is considered a threat to the ozone layer in the stratosphere. Calculate the root-mean-square velocity of Freon-12 molecules in the lower stratosphere where the temperature is −65°C.
A) 20 m/s
B) 120 m/s
C) 210 m/s
D) 260 m/s
E) 4.4 × 10 4 m/s
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the density of carbon dioxide gas at −25.2°C and 98.0 kPa?
A) 0.232 g/L
B) 0.279 g/L
C) 0.994 g/L
D) 1.74 g/L
E) 2.09 g/L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pressure of sulfur dioxide in a container is 159 kPa. What is this pressure in atmospheres?
A) 0.209 atm
B) 0.637 atm
C) 1.57 atm
D) 21.2 atm
E) 15900 atm
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
For an ideal gas, a plot of PV/nRT versus P gives a straight line with a positive slope.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Convert a gas pressure of 485 cmHg to atmospheres.
A) 0.64 atm
B) 33.0 atm
C) 6.38 atm
D) 5.50 atm
E) 6.46 atm
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the van der Waals equation for real gases to calculate the pressure exerted by 1.00 mole of ammonia at 27°C in a 750-mL container. (a = 4.17 L2·atm/mol2, b = 0.0371 L/mol)
A) 23.2 atm
B) 27.1 atm
C) 32.8 atm
D) 42.0 atm
E) 32.8 torr
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the conditions of STP?
A) 0 K and l atm
B) 273.15 K and 760 torr
C) 0°C and 760 atm
D) 273.15°C and 760 torr
E) None of these choices are correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ideal gas law tends to become inaccurate when
A) the pressure is lowered and molecular interactions become significant.
B) the pressure is raised and the temperature is lowered.
C) the temperature is raised above the temperature of STP.
D) large gas samples are involved.
E) the volume expands beyond the standard molar volume.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lithium oxide is an effective absorber of carbon dioxide and can be used to purify air in confined areas such as space vehicles. What volume of carbon dioxide can be absorbed by 1.00 kg of lithium oxide at 25°C and 1.00 atm? Li2O(aq) + CO2(g) → Li2CO3(s)
A) 687 mL
B) 819 mL
C) 687 L
D) 819 L
E) 22.4 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 0.750 L of argon at 1.50 atm and 177°C and 0.235 L of sulfur dioxide at 95.0 kPa and 63.0°C are added to a 1.00-L flask and the flask's temperature is adjusted to 25.0°C, what is the resulting pressure in the flask?
A) 0.0851 atm
B) 0.244 atm
C) 0.946 atm
D) 1.74 atm
E) 1.86 atm
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A flask containing helium gas is connected to an open-ended mercury manometer. The open end is exposed to the atmosphere, where the prevailing pressure is 752 torr. The mercury level in the open arm is 26 mm above that in the arm connected to the flask of helium. What is the helium pressure, in torr?
A) −26 torr
B) 26 torr
C) 726 torr
D) 778 torr
E) None of these choices are correct.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pressure in a 7.50-L flask if 0.15 mol of carbon dioxide is added to 0.33 mol of oxygen? The temperature of the mixture is 48.0°C.
A) 0.252 atm
B) 0.592 atm
C) 1.69 atm
D) 3.96 atm
E) 4.80 atm
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the lines on the figure below is the best representation of the relationship between the volume of a gas and its Celsius temperature, other factors remaining constant? 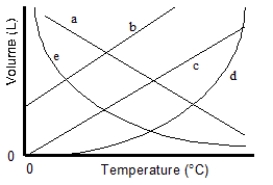
A) a
B) b
C) c
D) d
E) e
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compressed gas cylinder containing 1.50 mol methane has a volume of 3.30 L. What pressure does the methane exert on the walls of the cylinder if its temperature is 25°C?
A) 9.00 × 10 −2 atm
B) 0.933 atm
C) 1.11 atm
D) 1.70 atm
E) 11.1 atm
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of ammonia gas at 65.5°C and 524 torr has a volume of 15.31 L. What is its volume when the temperature is −15.8°C and its pressure is 524 torr?
A) 3.69 L
B) 11.6 L
C) 20.2 L
D) 63.5 L
E) Not possible, since the volume would have to be negative
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 98 of 98
Related Exams