A) Al(s) is oxidized and is the anode.
B) Al(s) is oxidized and is the cathode.
C) Cd(s) is oxidized and is the anode.
D) Cd(s) is oxidized and is the cathode.
E) No reaction occurs because E°cell < 0.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the half-reaction that occurs at the cathode during the electrolysis of an aqueous sodium iodide solution? 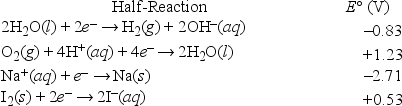
A) Na+ + e- →
B) O2(g) + 4H+(aq) + 4e- → 2H2O(l)
C) 2H2O + 2e- → H2 + 2OH-
D) I2 + 2e- → 2I-
E) 2I- → I2 + 2e-
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following standard reduction potentials in acid solution 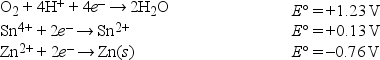 write the formula of the strongest reducing agent.
write the formula of the strongest reducing agent.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation is correct?
A) E°cell = E°anode - E°cathode
B) E°cell = E°cathode + E°anode
C) E°cell = E°cathode - E°anode
D) E°cell = E°cathode /E°anode
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the half-reaction at the anode in a lead storage battery?
A) Pb(s) + PbO2(s) + 4H+(aq) + 2SO42-(aq) → 2PbSO4(s) + 2H2O(l)
B) PbO2(s) + 4H+(aq) + 2SO42-(aq) + 2e- → PbSO4(s) + 2H2O(l)
C) Pb(s) + SO42-(aq) → PbSO4(s) + 2e-
D) Pb(s) → Pb(s) + 2e-
E) H2(g) → 2H+(aq) + 2e-
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is E°cell for the following reaction? 2Fe2+(aq) + Cd2+(aq) → 2Fe3+(aq) + Cd(s) 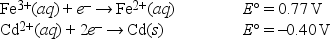
A) -0.37 V
B) 0.37 V
C) -1.17 V
D) 1.17 V
E) None of these choices is correct
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following balanced redox reaction. 3CuO(s) + 2NH3(aq) → N2(g) + 3H2O(l) + 3Cu(s) Which of the following statements is true?
A) CuO(s) is the oxidizing agent and copper is reduced.
B) CuO(s) is the oxidizing agent and copper is oxidized.
C) CuO(s) is the reducing agent and copper is oxidized.
D) CuO(s) is the reducing agent and copper is reduced.
E) CuO(s) is the oxidizing agent and N2(g) is the reducing agent.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation is correct?
A) E = -(RT/nF) lnK
B) E° = (RT/nF) lnK
C) E° = (RT/nF) lnQ
D) E = -(RT/nF) lnQ
E) E = E° + lnQ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
________ occurs at the cathode in a galvanic cell.
Correct Answer

verified
Correct Answer
verified
True/False
E > 0 and ΔG < 0 for a spontaneous process.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is meant by SHE?
A) Shared half electrodes
B) Shifting half of the electricity
C) Standard hydrogen electrode
D) Small helium electrode
E) Standard Hg electrode
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following standard reduction potentials, 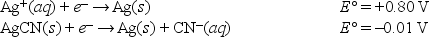 calculate the solubility product of AgCN at 25°C.
calculate the solubility product of AgCN at 25°C.
A) 4.3 × 10-14
B) 2.3 × 1013
C) 2.1 × 10-14
D) 5.1 × 1013
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the following electrochemical cell, what is the standard reduction potential of metal M at 298 K? (R = 8.314 J/K • mol, F = 96500 C/mol) 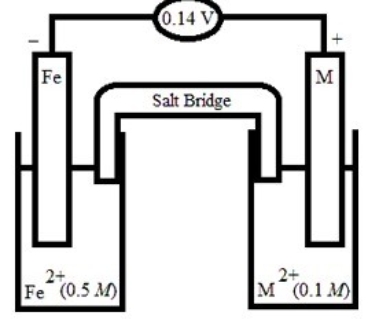
A) -0.54 V
B) +0.60 V
C) -0.30 V
D) +0.56 V
E) -0.28 V
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the data presented below, which is the strongest reducing agent? 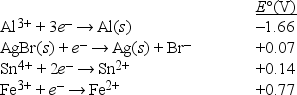
A) Fe3+
B) Fe2+
C) Br-
D) Al3+
E) Al(s)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain electrochemical cell has for its cell reaction: Zn + HgO → ZnO + Hg Which is the half-reaction occurring at the anode?
A) HgO + 2e- → Hg + O2-
B) Zn2++ 2e- → Zn
C) Zn → Zn2+ + 2e-
D) ZnO + 2e- → Zn
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following redox equation is balanced using the smallest whole-number coefficients, what is the coefficient of NO2? Sn + HNO3 → SnO2 + NO2 + H2O (acidic solution)
A) 1
B) 2
C) 3
D) 4
E) 5
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the purpose of a salt bridge in an electrochemical cell?
A) A salt bridge provides electrical contact between the electrodes that make up the cell.
B) A salt bridge carries electrons between the cathode and anode.
C) A salt bridge insulates the two electrodes.
D) A salt bridge allows the electrolyte solution to siphon from one side of the cell to the other so the levels remain equal.
E) A salt bridge allows electrolyte to flow from one half-cell to the other in order to maintain electroneutrality.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the equilibrium constant at 25°C for the following reaction? (R = 8.314 J/K • mol, F = 96,500 C • mol-1) 
A) 1.2 × 105
B) 1.4 × 1010
C) 8.6 × 10-6
D) 7.1 × 10-11
E) 2.3 × 1023
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the following equation is balanced with the lowest whole number coefficients, the coefficients are: Cu(s) + H+(aq) + NO3-(aq) → NO(g) + H2O(l) + Cu2+(aq)
A) 1, 2, 1, 1, 1, 1
B) 3, 2, 1, 1, 1, 3
C) 3, 2, 4, 2, 4, 3
D) 1, 8, 2, 2, 4, 1
E) 3, 8, 2, 2, 4, 3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
A SHE has the acid concentration of 1 M and the H2 pressure is 1 atm.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 122
Related Exams