A) tetrahedral
B) trigonal planar
C) trigonal pyramidal
D) trigonal bipyramidal
E) seesaw
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
In not more than two sentences, explain when and why chemists make use of the concept of hybridization.
Correct Answer

verified
Chemists postulate hybridizati...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
According to molecular orbital theory, what is the bond order in the O2+ ion?
A) 5.5
B) 5
C) 4
D) 2.5
E) 1.5
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most reasonable prediction for the three F-Br-F bond angles in BrF3?
A) 90°, 90°, and 180°
B) 86°, 94°, and 180°
C) 86°, 86°, and 172°
D) 94°, 94°, and 172°
E) 120°, 120°, and 120°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
The number of lone pairs on the central atom is used to determine the ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a homonuclear diatomic molecule, which molecular orbital is the highest in energy?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) All of these orbitals have the same energy.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using the VSEPR model, what is the predicted molecular geometry of the PCl3 molecule?
A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal
E) tetrahedral
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
How many electron domains are on the central atom for a sp3d hybridized atom?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, the triple bond in ethyne (acetylene, C2H2) consists of
A) three σ bonds and no ![]() bonds.
bonds.
B) two σ bonds and one ![]() bond.
bond.
C) one σ bond and two ![]() bonds.
bonds.
D) no σ bonds and three ![]() bonds.
bonds.
E) one σ bond, one ![]() bond, and one bond intermediate between σ and
bond, and one bond intermediate between σ and ![]() .
.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the bond order for the species O2−?
A) 1
B) 1.5
C) 2
D) 2.5
E) None of these choices.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules is polar?
A) PBr5
B) CCl4
C) BrF5
D) XeF2
E) XeF4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
According to molecular orbital theory, all diatomic molecules with an even number of electrons are diamagnetic.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which figure best illustrates the hybrid orbitals on iodine in ICl2-?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many bonds are there in one molecule of acrylonitrile (shown below) ? 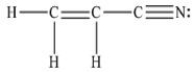
A) 1
B) 2
C) 3
D) 5
E) 9
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct hybrid orbital diagram representing the hybridization of iodine in ICl5?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules is polar?
A) CH4
B) CHBr3
C) F2
D) CBr4
E) CO2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules has a zero dipole moment?
A) CO
B) CH2Cl2
C) SO3
D) SO2
E) NH3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is true concerning the dipole moment of a CO2 molecule?
A) The dipole moment is zero because the C-O bond is nonpolar.
B) The dipole moment is zero because the molecule is linear.
C) The dipole moment is zero because the molecule is bent.
D) The dipole moment is nonzero because the molecule is linear.
E) The dipole moment is nonzero because the molecule is bent.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For a polyatomic ion having the general formula AB3n±, where A is an atom from Group 6A, B is an atom from Group 7A, and the ionic charge is n±, what charge must the ion have in order to yield a molecular geometry that is trigonal pyramidal?
A) 3-
B) 1-
C) 1+
D) 3+
E) 5+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model, the predicted molecular geometry of ammonia, NH3, is
A) linear.
B) trigonal planar.
C) bent.
D) tetrahedral.
E) trigonal pyramidal.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 139
Related Exams