A) the frozen form is more dense than the liquid form
B) the frozen form is less dense than the liquid form
C) water is a solvent
D) water has a high heat capacity
E) water has a high heat of evaporation
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is NOT true about covalent bonds?
A) Covalent bonds form when an electron is completely lost or gained from an atom.
B) A covalent molecule contains one or more covalent bonds.
C) A single covalent bond is drawn as a line between two atoms.
D) A pair of electrons is shared between two atoms for each covalent bond.
E) Shared electrons allow an atom to complete its outer electron shell in a covalent molecule.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the atomic number of an element is 6 and the atomic mass is 12.01,how many protons are there in the nucleus?
A) 12
B) 6
C) 24
D) 52
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Not Answered
Draw several (5 or 6)individual,unbonded water molecules.Simulate what happens when table salt (Na+Cl-)is added to water.Use the model you created to explain why salt is added to the roads in a 'snowy',cold climate.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If you place the corner of a paper towel into a droplet of water the water moves across the paper towel.Which of the following would explain the movement of the water?
A) surface tension
B) cohesion
C) adhesion
D) both cohesion and adhesion
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the maximum number of electrons that will be in the 1st valence shell?
A) 2
B) 1
C) 3
D) 6
E) 8
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many elements are required to form a molecule?
A) at least 2
B) at least 3
C) at least 4
D) at least 5
E) only 1
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is NOT true about polar covalent bonds?
A) Most covalent bonds are nonpolar,with electrons shared fairly equally between the atoms.
B) Polar covalent bonds are important in the characteristics of water.
C) Electrons are shared unequally in a polar covalent bond.
D) The larger atom in a polar bond attracts the electron more strongly than the smaller atom.
E) The oxygen of a water molecule is electropositive relative to the hydrogen.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If an element contains 8 electrons how many electrons will be placed in the 2nd valence shell?
A) 6
B) 2
C) 8
D) 5
E) 11
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be a proposed mechanism by which stomach antacids work?
A) Antacids dilute the solution,therefore lowering the pH.
B) Antacids are bases and by definition can absorb H+ out of a solution.
C) Antacids are bases and by definition can absorb OH- out of a solution.
D) Antacids contain mostly water and so they neutralize the solution.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Not Answered
Following nitrogen (78%)and oxygen (21%),argon is the next most common gas in the atmosphere (less than 1%).Checking the table of elements,you discover that argon is one of a family of atoms with outer shells already full of electrons.How is this related to the fact that these atoms have virtually no biological importance?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is NOT true about subatomic particles?
A) Protons are found in the nucleus.
B) Neutrons have no electrical charge.
C) Electrons contain much less mass than neutrons.
D) Electrons are found in orbitals around the nucleus.
E) All electrons in an atom contain the same amount of energy.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The scale ranges from 1 to 15.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which property of water allows it to act as a transport medium?
A) cohesion
B) high heat of evaporation
C) high heat capacity
D) water is solvent
E) the frozen form is less dense than the liquid form
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following concept circles best depicts the relationship between molecules and compounds (c = compound and m = molecule) . A.
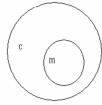 ?.
?.
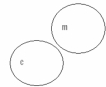 C.
C.
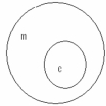
A) Option A
B) Option B
C) Option C Incorrect
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substances are on the basic side of the pH scale?
A) baking soda,oven cleaner & human blood
B) baking soda,oven cleaner & urine
C) tomatoes,oven cleaner & human blood
D) beer,vinegar & black coffee
E) Great Salt Lake,oven cleaner,tears
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is NOT true about the electrical charges in chemistry?
A) Protons carry a positive charge.
B) In an atom,the number of protons and neutrons must be equal.
C) An atom is neutral when the positive and negative charges balance.
D) An ion contains one or more positive or negative charges.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 57 of 57
Related Exams