A) linear
B) bent
C) trigonal planar
D) trigonal pyramidal
E) tetrahedral
G) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
In which of the following compounds will the molecules not form hydrogen bonds with each other? 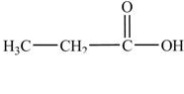
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following in order of decreasing electronegativity. Mg Cl F
A) Mg > Cl > F
B) F > Cl > Mg
C) Cl > F > Mg
D) Cl > Mg > F
E) F > Mg > Cl
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model, the predicted molecular geometry of ammonia, NH3, is
A) linear.
B) trigonal planar.
C) bent.
D) tetrahedral.
E) trigonal pyramidal.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of ClF2− as predicted by the VSEPR model?
A) linear
B) bent
C) seesaw
D) T-shaped
E) trigonal bipyramidal
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the molecular geometry and polarity of the SO2 molecule.
A) linear, polar
B) linear, nonpolar
C) bent, polar
D) bent, nonpolar
E) trigonal planar, polar
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane is a component of fuel used in internal combustion engines.The dominant intermolecular forces in octane are
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) covalent bonds.
E) carbon-hydrogen bonds.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which kinds of intermolecular forces exist between propane molecules? I.dispersion forces II) dipole-dipole interactions Ion-dipole interactions
A) I only
B) II only
C) III only
D) I, II, and III
E) I and III
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following molecules is nonpolar?
A) NH3
B) OF2
C) CH3Cl
D) H2O
E) BeCl2
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between pentane (C5H12) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) carbon-carbon bonds.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is defined as the attractive forces between polar molecules? I.dispersion forces II) dipole-dipole interactions Ion-dipole interactions
A) I and III
B) II only
C) III only
D) I, II, and III
E) I only
G) C) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
How many lone pairs of electrons need to be added to complete this Lewis structure? 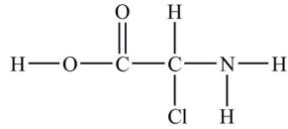
A) 5
B) 8
C) 6
D) 1
E) 16
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pure substances has the strongest dispersion forces?
A) C4H10
B) C5H12
C) C6H14
D) C7H16
E) C8H18
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following in order of increasing electronegativity. Se K Cl
A) Cl < Se < K
B) K < Se < Cl
C) K < Cl < Se
D) Se < Cl < K
E) Cl < K < Se
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Place the following in order of increasing electronegativity. Cs O S
A) O < S < Cs
B) S < O < Cs
C) Cs < S < O
D) Cs < O < S
E) O < Cs < S
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pure substances will exhibit hydrogen bonding?
A) CH4
B) H2C=CH2
C) H2O
D) CHCl3
E) CHF3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of resonance structures for the nitrate ion that satisfy the octet rule is
A) 1.
B) 2.
C) 3.
D) 4.
E) none of these
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model, the predicted molecular geometry of SiCl4 is
A) linear.
B) trigonal planar.
C) bent.
D) tetrahedral.
E) trigonal pyramidal.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of N2O as predicted by the VSEPR model? 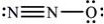
A) trigonal pyramidal
B) trigonal planar
C) tetrahedral
D) bent
E) linear
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following pure substance has both dispersion forces and dipole-dipole forces?
A) HCl
B) BCl3
C) Br2
D) H2
E) CO2
G) A) and C)
Correct Answer

verified
A
Correct Answer
verified
Showing 1 - 20 of 71
Related Exams