A) Increase the pressure of H2 to 2.0 atm.
B) Decrease the mass of the nickel electrode.
C) Lower the pH of the cell electrolyte.
D) Decrease the concentration of Ni2+ ion.
E) None of the above.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these metals will not reduce water to hydrogen in basic solution under standard conditions?
A) Cd
B) Sr
C) Mg
D) Ba
E) K
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the minimum voltage required for the electrolysis of 1.0 M NaCl in neutral solution. 2H2O + 2Cl- (1.0 M) H2(1 atm) + Cl2(1 atm) + 2OH- (1 * 10-7 M)
A) 2.19 V
B) 1.78 V
C) 0.41 V
D) -0.41 V
E) -1.78 V
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these metals will reduce water to hydrogen in basic solution under standard conditions?
A) Pb
B) Fe
C) Zn
D) Na
E) Ag
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The overall reaction 2Co3+(aq) + 2Cl-(aq) 2Co2+(aq) + Cl2(g) has the standard cell voltage E°cell = 0.46 V. Given E° = 1.36 V for the reaction Cl2(g) + 2e- 2Cl-(aq) , calculate the standard reduction potential for the following the half reaction at 25°C: Co3+ + e- Co2+
A) 1.82 V
B) -0.90 V
C) 0.90 V
D) -1.82 V
E) -1.36 V
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell based on the spontaneous reaction 2AgCl(s) + Zn(s) 2Ag(s) + 2Cl- + Zn2+. If the zinc ion concentration is kept constant at 1 M, and the chlorine ion concentration is decreased from 1 M to 0.001 M, the cell voltage should
A) increase by 0.06 V.
B) increase by 0.18 V.
C) decrease by 0.06 V.
D) decrease by 0.18 V.
E) increase by 0.35 V.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation using the set of smallest whole-numbers coefficients. What is the sum of the coefficients? HI + HNO3 I2 + NO (acidic solution)
A) 5
B) 7
C) 14
D) 17
E) None of these.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider an electrochemical cell with the following cell reaction where all reactants and products are at standard-state conditions: Cu2+(aq) + H2(g) Cu(s) + 2H+(aq) . Predict the effect on the emf of this cell of adding NaOH solution to the hydrogen half-cell until the pH equals 7.0.
A) The emf will increase.
B) The emf will decrease.
C) No change in the emf will be observed.
E) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
When a solution of a certain gadolinium salt is electrolyzed with a current of 1.0 A for 2.0 h, 0.025 mol of Gd metal forms. Calculate the charge on the gadolinium ion in the salt.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the equilibrium constant (Keq) at 25°C for the reaction Cl2(g) + 2Br- (aq) 2Cl- (aq) + Br2(l)
A) 1.5 * 10-10
B) 6.3 * 109
C) 1.3 * 1041
D) 8.1 * 104
E) 9.8
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following notation for an electrochemical cell Pt(s) | H2(g) | H+(aq) || Ag+(aq) | Ag(s) What is the balanced overall (net) cell reaction?
A) 2H+(aq) + 2Ag+(aq) H2(g) + 2Ag(s)
B) H2(g) + 2Ag(s) H+(aq) + 2Ag+(aq)
C) 2H+(aq) + 2Ag(s) H2(g) + 2Ag+(aq)
D) H2(g) + Ag+(aq) H+(aq) + Ag(s)
E) H2(g) + 2Ag+(aq) 2H+(aq) + 2Ag(s)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following reagents is capable of transforming Br- (aq) to Br2(l) under standard-state conditions?
A) I- (aq)
B) NO3- (aq)
C) Ag+ (aq)
D) Al3+ (aq)
E) Au3+ (aq)
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following standard reduction potentials in acid solution O2 + 4H+ + 4e- 2H2O E° = +1.23 V Sn4+ + 2e- Sn2+ E° = +0.13 V Zn2+ + 2e- Zn(s)E° = -0.76 V write the formula of the strongest oxidizing agent.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using a table of standard reduction potentials, determine which of these reactions (if any) is/are nonspontaneous in the direction indicated at 25°C.
A) 2Fe3+ + 2Cl- 2Fe2+ + Cl2(g)
B) 2Fe3+ + 2Br- 2Fe2+ + Br2(l)
C) 2Fe3+ + 2I- 2Fe2+ + I2(s)
D) A and B
E) All are spontaneous.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following standard reduction potentials in acid solution: 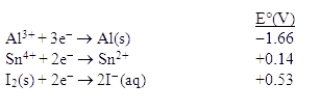 Which is the weakest oxidizing agent in this list?
Which is the weakest oxidizing agent in this list?
A) Al3+(aq)
B) Al(s)
C) I-(aq)
D) I2(s)
E) Sn4+(aq)
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the equilibrium constant, Keq, at 25°C for the reaction 2Br- (aq) + I2(s) Br2(l) + 2I- (aq)
A) 5.7 * 10-19
B) 18.30
C) 1.7 * 1054
D) 1.9 * 1018
E) 5.7 * 10-55
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Calculate the cell emf for the following reaction at 25°C: 2Ag+(0.010 M)+ H2(1 atm) 2Ag(s)+ 2H+(pH = 6.0)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete and balance the following redox equation. What is the coefficient of H2O when the equation is balanced using the set of smallest whole-number coefficients? MnO4- + SO32- Mn2+ + SO42- (acidic solution)
A) 3
B) 4
C) 5
D) 8
E) None of these.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Will H2(g)form when Fe is placed in 1.0 M HCl?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain electrochemical cell has for its cell reaction: Zn + HgO ZnO + Hg Which is the half-reaction occurring at the anode?
A) HgO + 2e- Hg + O2-
B) Zn2+ + 2e- Zn
C) Zn Zn2+ + 2e-
D) ZnO + 2e- Zn
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 138
Related Exams