A) 0) 576 kJ
B) 12.2 kJ
C) 38.4 kJ
D) 52.9 kJ
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Shown is a concentration versus time plot for a reaction involving gases A,B,and C. 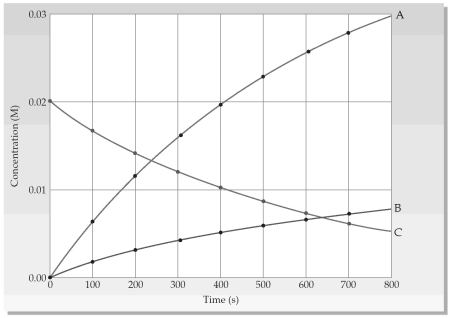 -Which equation best represents the reaction?
-Which equation best represents the reaction?
A) 4A(g) → B(g) + 2C(g)
B) 4A(g) + B(g) → 2C(g)
C) 2C(g) → 4A(g) + B(g)
D) 2C(g) + B(g) → 4A(g)
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a reaction that occurs by the following mechanism:
A + BC → AC + B
AC + D → A + CD
The potential energy profile for this reaction is shown below. 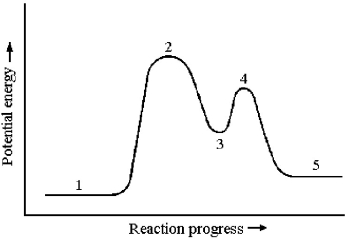 -Transition states occur at which reaction stages?
-Transition states occur at which reaction stages?
A) reaction stages 1 and 5
B) reaction stages 2,3,and 4
C) reaction stages 2 and 4
D) reaction stage 3
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The rate constant,k,for a first-order reaction is equal to 4.2 × 10-4 s-1.What is the half-life for the reaction?
A) 2) 9 × 10-4 s
B) 1) 2 × 103 s
C) 1) 7 × 103 s
D) 2) 4 × 103 s
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the minimum energy barrier that must be overcome for a chemical reaction to occur?
A) activation energy
B) net energy
C) potential energy
D) rate limiting energy
F) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
A gaseous reaction occurs by a two-step mechanism,shown below. Step 1: AX +Y2 → AXY2 Step 2: AXY2 + AX → 2 AXY If the rate law for this reaction is Rate = k[AX]2[Y2],the intermediate is ________,and step ________ is the rate-determining step.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of dinitrogen pentoxide is described by the chemical equation 2 N2O5(g) → 4 NO2(g) + O2(g) If the rate of appearance of NO2 is equal to 0.560 mol/min at a particular moment,what is the rate of appearance of O2 at that moment?
A) 0) 140 mol/min
B) 0) 280 mol/min
C) 1) 12 mol/min
D) 2) 24 mol/min
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following reaction is first order in A and first order in B:
A + B → Products Rate = k[A][B]
What is the rate constant k of this reaction in vessel (b) relative to the rate constant k of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules. ![The following reaction is first order in A and first order in B: A + B → Products Rate = k[A][B] What is the rate constant k of this reaction in vessel (b) relative to the rate constant k of this reaction in vessel (a) ? Each vessel has the same volume.Shaded spheres represent A molecules,and unshaded spheres represent B molecules. A) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:2 B) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:1 C) rate constant k in vessel (b) /rate constant k in vessel (a) = 2:1 D) rate constant k in vessel (b) /rate constant k in vessel (a) = 4:1](https://d2lvgg3v3hfg70.cloudfront.net/TB4939/11ea7a38_c94c_c424_aa4c_2dd2aedcd5e5_TB4939_00.jpg)
A) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:2
B) rate constant k in vessel (b) /rate constant k in vessel (a) = 1:1
C) rate constant k in vessel (b) /rate constant k in vessel (a) = 2:1
D) rate constant k in vessel (b) /rate constant k in vessel (a) = 4:1
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of dinitrogen pentoxide is described by the chemical equation 2 N2O5(g) → 4 NO2(g) + O2(g) If the rate of disappearance of N2O5 is equal to 1.40 mol/min at a particular moment,what is the rate of appearance of NO2 at that moment?
A) 0) 700 mol/min
B) 1) 40 mol/min
C) 2) 80 mol/min
D) 5) 60 mol/min
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The aquation of tris(1,10-phenanthroline) iron(II) in acid solution takes place according to the equation: Fe(phen) 32+ + 3 H3O+ + 3 H2O → Fe(H2O) 62+ + 3 phenH+. If the activation energy,Ea,is 126 kJ/mol and the rate constant at 30°C is 9.8 × 10-3 min-1,what is the rate constant at 35°C?
A) 4) 4 × 10- 3 min-1
B) 2) 2 × 10- 2 min-1
C) 4) 5 × 101 min-1
D) 2) 3 × 102 min-1
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Given the following hypothetical reaction: 2 E(g) + F(g) + G(g) → products. If the rate law is: Rate = k[E]2[F]-1,what is the overall order of reaction?
A) zero
B) first
C) second
D) third
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second-order reaction 2 Mn(CO) 5 → Mn2(CO) 10,has a rate constant equal to 3) 0 × 109 M-1 s-1 at 25°C.If the initial concentration of Mn(CO) 5 is 2.0 × 10-5 M,how long will it take for 90.% of the reactant to disappear?
A) 6) 7 × 10- 16 s
B) 7) 4 × 10- 15 s
C) 1) 5 × 10- 4 s
D) 6) 0 × 103 s
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first-order decomposition of hydrogen peroxide occurs according to the equation 2 H2O2(aq) → 2 H2O(l) + O2(g) Using data from a concentration-time study of this reaction,which plot will produce a straight line?
A) [H2O2] versus time
B) [H2O2]2 versus time
C) 1/[H2O2] versus time
D) ln[H2O2] versus time
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the units for rate are M s-1,what are the units for the rate constant,k,if the overall order of the reaction is three?
A) s-1
B) M2 s-1
C) M-1 s-1
D) M-2 s-1
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the first-order decomposition of A molecules (shaded spheres) in two vessels of equal volume.How will the rate of decomposition in vessel (a) be affected if the volume of the vessel is decreased by a factor of 2? 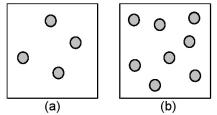
A) decrease by 1/2
B) increase by 2
C) increase by 4
D) stay the same
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decay curve shown below approximates the decay of phosphorus-32.What is the approximate half-life of phosphorus-32? 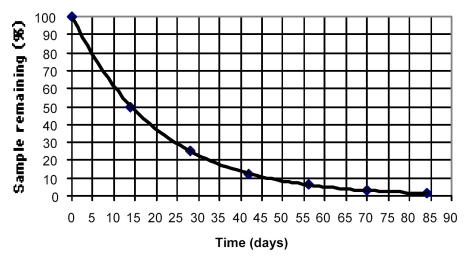
A) 14 days
B) 27 days
C) 35 days
D) 37.5 days
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Shown is a concentration versus time plot for a reaction involving gases A,B,and C. ![Shown is a concentration versus time plot for a reaction involving gases A,B,and C. -Over the time interval 300 to 400 seconds,the rate of reaction with respect to A is Δ[A]/Δt = 3.7 × 10<sup>-5</sup> M/s.Over the same time interval what is the rate of reaction with respect to B,Δ[B]/Δt? A) Δ[B]/Δt = Δ[A]/Δt = 3.7 × 10<sup>-5</sup> M/s B) Δ[B]/Δt = (1/4) (Δ[A]/Δt) = (1/4) (3.7 × 10<sup>-5</sup> M/s) = 9.2 × 10<sup>-6</sup> M/s C) Δ[B]/Δt = (1/2) (Δ[A]/Δt) = (1/2) (3.7 × 10<sup>-5</sup> M/s) = 1.8 × 10<sup>-5</sup> M/s D) Δ[B]/Δt = -(1/2) (Δ[A]/Δt) = -(1/2) (3.7 × 10<sup>-5</sup> M/s) = -1.8 × 10<sup>-5 </sup>M/s](https://d2lvgg3v3hfg70.cloudfront.net/TB4939/11ea7a38_c94b_8b9f_aa4c_d5d6a940b43a_TB4939_00_TB4939_00_TB4939_00_TB4939_00_TB4939_00.jpg) -Over the time interval 300 to 400 seconds,the rate of reaction with respect to A is Δ[A]/Δt = 3.7 × 10-5 M/s.Over the same time interval what is the rate of reaction with respect to B,Δ[B]/Δt?
-Over the time interval 300 to 400 seconds,the rate of reaction with respect to A is Δ[A]/Δt = 3.7 × 10-5 M/s.Over the same time interval what is the rate of reaction with respect to B,Δ[B]/Δt?
A) Δ[B]/Δt = Δ[A]/Δt = 3.7 × 10-5 M/s
B) Δ[B]/Δt = (1/4) (Δ[A]/Δt) = (1/4) (3.7 × 10-5 M/s) = 9.2 × 10-6 M/s
C) Δ[B]/Δt = (1/2) (Δ[A]/Δt) = (1/2) (3.7 × 10-5 M/s) = 1.8 × 10-5 M/s
D) Δ[B]/Δt = -(1/2) (Δ[A]/Δt) = -(1/2) (3.7 × 10-5 M/s) = -1.8 × 10-5 M/s
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Haber process is the synthesis of ammonia gas from hydrogen and nitrogen on a hot metal surface.What is the catalyst and what type of catalysis is occurring?
A) H2,homogeneous
B) N2,homogeneous
C) NH3,homogeneous
D) metal surface,heterogeneous
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
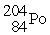 Decays by electron capture to give
Decays by electron capture to give 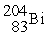 If unshaded spheres represent
If unshaded spheres represent 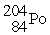 Atoms and shaded spheres represent
Atoms and shaded spheres represent 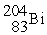 Atoms,how many half-lives have passed in the following example?
Atoms,how many half-lives have passed in the following example? 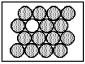
A) 1 half-life
B) 2 half-lives
C) 3 half-lives
D) 4 half-lives
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In aqueous solution,hypobromite ion,BrO-,reacts to produce bromate ion,BrO3-,and bromide ion,Br-,according to the following chemical equation. ![In aqueous solution,hypobromite ion,BrO<sup>-</sup>,reacts to produce bromate ion,BrO<sub>3</sub><sup>-</sup>,and bromide ion,Br<sup>-</sup>,according to the following chemical equation. A plot of 1/[BrO<sup>-</sup>] vs.time is linear and the slope is equal to 0.056 M<sup>-1</sup>s<sup>-1</sup>.If the initial concentration of BrO<sup>-</sup> is 0.80 M,how long will it take one-half of the BrO<sup>- </sup>ion to react? A) 4) 5 × 10<sup>-2</sup> s B) 7) 1 s C) 12 s D) 22 s](https://d2lvgg3v3hfg70.cloudfront.net/TB4939/11ea7a38_c952_b7c3_aa4c_290845be0aeb_TB4939_00.jpg) A plot of 1/[BrO-] vs.time is linear and the slope is equal to 0.056 M-1s-1.If the initial concentration of BrO- is 0.80 M,how long will it take one-half of the BrO- ion to react?
A plot of 1/[BrO-] vs.time is linear and the slope is equal to 0.056 M-1s-1.If the initial concentration of BrO- is 0.80 M,how long will it take one-half of the BrO- ion to react?
A) 4) 5 × 10-2 s
B) 7) 1 s
C) 12 s
D) 22 s
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 206
Related Exams