A) 0.619 s
B) 124 s
C) 387 s
D) 429 s
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement below regarding the half-life of a zeroth-order reaction is true?
A) Each half-life is half as long as the preceding half-life.
B) Each half-life is twice as long as the preceding half-life.
C) Each half-life is four times as long as the preceding half-life.
D) The half-life remains unchanged throughout the course of the reaction.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which part of the Arrhenius equation contains a term which measures the number of molecules that have the correct orientation for reaction?
A) activation energy
B) e-Ea/RT
C) frequency factor
D) none of these
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction shown below,what is the order of reaction with respect to Br? ClO2-(aq) + 4 Br-(aq) + 4 H+(aq) → Cl-(aq) + 2 Br2(aq) +2 H2O(l)
A) 1st order
B) 2nd order
C) 4th order
D) can't tell from the information provided
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of ammonia to nitrogen and hydrogen on a tungsten filament at 800°C is independent of the concentration of ammonia at high pressures of ammonia.What is the order of the reaction with respect to ammonia?
A) zero
B) first
C) second
D) third
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following set of data was obtained by the method of initial rates for the reaction: S2O82-(aq) + 3 I-(aq) → 2 SO42-(aq) + I3-(aq)
What is the rate law for the reaction? ![The following set of data was obtained by the method of initial rates for the reaction: S<sub>2</sub>O<sub>8</sub><sup>2-</sup>(aq) + 3 I<sup>-</sup>(aq) → 2 SO<sub>4</sub><sup>2-</sup>(aq) + I<sub>3</sub><sup>-</sup>(aq) What is the rate law for the reaction? A) Rate = k[S<sub>2</sub>O<sub>8</sub><sup>2-</sup>][I<sup>-</sup>] B) Rate = k[S<sub>2</sub>O<sub>8</sub><sup>2-</sup>][I<sup>-</sup>]<sup>5</sup> C) Rate = k[S<sub>2</sub>O<sub>8</sub><sup>2-</sup>][I<sup>-</sup>]<sup>2</sup> D) Rate = k[S<sub>2</sub>O<sub>8</sub><sup>2-</sup>]<sup>2</sup>[I<sup>-</sup>]](https://d2lvgg3v3hfg70.cloudfront.net/TB4940/11ea7e2d_d153_0b47_a2f7_471b28009a8b_TB4940_00.jpg)
A) Rate = k[S2O82-][I-]
B) Rate = k[S2O82-][I-]5
C) Rate = k[S2O82-][I-]2
D) Rate = k[S2O82-]2[I-]
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
The reaction below is second order in ClO2 and first order in OH-.When the concentration of ClO2 is 0.020 M and the concentration of OH- is 0.10 M,the initial rate of reaction is 8.00 × 10-4 M/s.What is the rate constant,k,for this reaction? 2 ClO2(aq)+ 2 OH-(aq)→ ClO3-(aq)+ ClO2-(aq)+ H2O(l)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Acetaldehyde decomposes at 750 K: CH3CHO → CO + CH4.The reaction is first order in acetaldehyde and the half-life of the reaction is found to be 530 seconds.What is the rate constant for the reaction at this temperature?
A) 2.7 × 10-3 s-1
B) 1.3 × 10-3 s-1
C) 2.7 × 102 s-1
D) 7.6 × 102 s-1
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following set of data was obtained by the method of initial rates for the reaction: (H3C) 3CBr + OH- → (H3C) 3COH + Br-
What is the order of reaction with respect to ion,OH-? 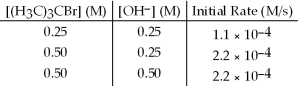
A) zero
B) first
C) second
D) none of these
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the first-order decomposition of A molecules (shaded spheres) in two vessels of equal volume.How will the half-life of decomposition in vessel (a) be affected if the volume of the vessel is decreased by a factor of 2? 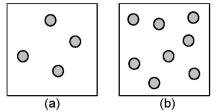
A) decrease by 1/2
B) increase by 2
C) increase by 4
D) stay the same
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The second-order reaction 2 Mn(CO) 5 → Mn2(CO) 10,has a rate constant equal to 3.0 × 109 M-1 s-1 at 25°C.If the initial concentration of Mn(CO) 5 is 5.0 × 10-5 M,how long will it take for 90.% of the reactant to disappear?
A) 1.7 × 10-15 s
B) 1.9 × 10-14 s
C) 6.0 × 10-5 s
D) 1.5 × 102 s
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The first-order reaction,SO2Cl2 → SO2 + Cl2,has a rate constant equal to 2.20 × 10-5 s-1 at 593 K.What percentage of the initial amount SO2Cl2 will remain after 3.00 hours?
A) 1.00%
B) 21.1%
C) 23.5%
D) 78.9%
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An aqueous reaction occurs by a two-step mechanism,shown below. Step 1: A2X2 + Y → A2X + XY Step 2: A2X2 + XY → A2X + X2 + Y What is the catalyst in this reaction?
A) A2X2
B) X2
C) XY
D) Y
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A gaseous reaction occurs by a two-step mechanism,shown below. Step 1: AX +Y2 ⇌ AXY2 fast Step 2: AXY2 + AX → 2 AXY slow Including concentration of only reactants and products,what is the rate law for this reaction?
A) Rate = k[AX][XY2]
B) Rate = k[AXY2]/[AX][Y2]
C) Rate = k[AX]2[Y2]
D) Rate = k[AXY]2/[A Y2][AX]
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The reaction that occurs in a Breathalyzer,a device used to determine the alcohol level in a person's bloodstream,is given below.If the rate of appearance of Cr2(SO4) 3 is 1.24 mol/min at a particular moment,what is the rate of disappearance of C2H6O at that moment? 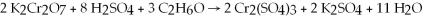
A) 0.413 mol/min
B) 0.826 mol/min
C) 1.86 mol/min
D) 3.72 mol/min
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The decomposition of hydrogen peroxide is given by the following reaction: 2 H2O2(aq) → 2 H2O(l) + O2(g) In the presence of KI the reaction is thought to occur by the following mechanism: Step 1: H2O2 + I- → H2O + IO- Step 2: IO- + H2O2 → H2O + O2 + I- What is the role of I- in this mechanism?
A) catalyst
B) frequency factor
C) product
D) transition state
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider a reaction that occurs by the following one-step mechanism:
A2 + B2 → 2 AB
The potential energy profile for this reaction is shown below. 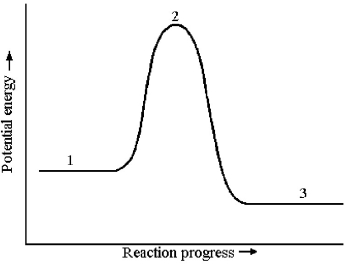 -What is the species present at reaction stage 2?
-What is the species present at reaction stage 2?
A) an intermediate
B) a product
C) a reactant
D) a transition state
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The relative initial rates of the reaction A2 + B2 → products in vessels (a) -(d) are 1:1:4:4.Unshaded spheres represent A2 molecules,and shaded spheres represent B2 molecules present at the beginning of the reaction. 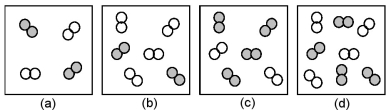 -What is the order of reaction with respect to B2?
-What is the order of reaction with respect to B2?
A) 0
B) 1
C) 2
D) 3
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements are true about reaction mechanisms? I.A rate law can be written from the molecularity of the slowest elementary step. II.The final rate law can include intermediates. III.The rate of the reaction is dependent on the fastest step in the mechanism. IV.A mechanism can never be proven to be the correct pathway for a reaction.
A) I,II,III
B) II,IV
C) I,III
D) I,IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
The decomposition of hydrogen peroxide occurs according to the equation 2 H2O2(aq)→ 2 H2O(l)+ O2(g) A concentration-time study of this reaction produces a straight line when ln[H2O2] is plotted versus time.Therefore,this is a ________ order reaction.
Correct Answer

verified
Correct Answer
verified
Showing 121 - 140 of 190
Related Exams