A) Si
B) Se
C) S
D) Sc
E) Sr
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Covalent bonding typically occurs when a ______________ bonds with a ______________.
Correct Answer

verified
non-metal;...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the following period 3 chlorides would be expected to have the highest melting point?
A) MgCl2
B) AlCl3
C) SiCl4
D) PCl3
E) SCl2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The melting points of metals are only moderately high because
A) metallic bonding is weak.
B) metals have fewer bonding electrons than non-metals.
C) metals also have relatively low boiling points.
D) the melting process does not break the metallic bonds.
E) metals prefer to be bonded to non-metals.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Using appropriate, real examples to illustrate your answer, describe the correlation between bond energy and bond length for a series of varying bond order.
Correct Answer

verified
Carbon and oxygen form single, double, a...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
When an atom is represented in a Lewis electron dot symbol, the element symbol represents ______________ and the dots represent ______________.
Correct Answer

verified
nucleus an...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The lattice energy for ionic crystals increases as the charge on the ions _____________ and the size of the ions __________________.
A) increases, increases
B) increases, decreases
C) decreases, increases
D) decreases, decreases
E) None of the above is generally correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the most polar bond amongst the following.
A) C-O
B) Si-F
C) Cl-F
D) C-F
E) C-I
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following contains ionic bonding?
A) CO
B) SrF2
C) Al
D) OCl2
E) HCl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Ionic bonding typically occurs when a ______________ bonds with a ______________.
Correct Answer

verified
Correct Answer
verified
True/False
The electrostatic energy of two charged particles is inversely proportional to the distance between them.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements is the most electronegative?
A) Ne
B) Rb
C) P
D) I
E) Cl
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a covalent compound?
A) Na2O
B) CaCl2
C) Cl2O
D) CsCl
E) Al2O3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogenation of double and triple bonds is an important industrial process. Calculate (in kJ) the standard enthalpy change H° for the hydrogenation of ethyne (acetylene) to ethane. 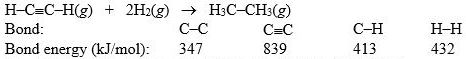
A) -296 kJ
B) -51 kJ
C) 51 kJ
D) 296 kJ
E) 381 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 74 of 74
Related Exams