A) C2H6
B) CH3OCH3
C) NO2
D) H2S
E) Ca(NO3) 2
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The strongest intermolecular interactions between ethyl alcohol (CH3CH2OH) molecules arise from
A) dipole-dipole forces.
B) London dispersion forces.
C) hydrogen bonding.
D) ion-dipole interactions.
E) carbon-oxygen bonds.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is the phase diagram for sulfur.Which phase has the highest density at 119°C? 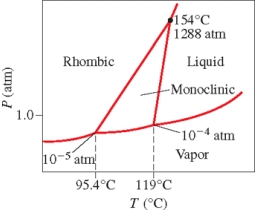
A) liquid
B) vapor
C) rhombic solid
D) monoclinic solid
E) liquid-vapor boundary line
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pure substances has the highest boiling point?
A) CH4
B) Cl2
C) Kr
D) CH3Cl
E) N2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
___________ ____________ are described by an instantaneous dipole.
Correct Answer

verified
London for...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Poor conductor of heat and electricity,soft,and low melting point are properties which describe a(n)_______________ crystal.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which kinds of intermolecular forces exist between propane molecules? I.dispersion forces II) dipole-dipole interactions III) ion-dipole interactions
A) I only
B) II only
C) III only
D) I,II,and III
E) I and III
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Only molecules which do not have dipole moments can experience dispersion forces.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
_______________ ________ are solids that lack a regular three-dimensional arrangement of atoms.
Correct Answer

verified
Correct Answer
verified
True/False
The shape of the water-to-glass meniscus results from the strong adhesive forces between glass and water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the response that lists the member of each of the following pairs that has the higher boiling point. I.H2O or KI,II.HF or HI,III.Cl2 or Br2
A) H2O,HF,and Cl2
B) KI,HF,and Br2
C) KI,HI,and Br2
D) H2O,HI,and Cl2
E) KI,HF,and Cl2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which substance has the lowest vapor pressure at room temperature?
A) HF
B) HCl
C) HBr
D) HI
E) H2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Variable melting point,variable hardness,and good conductor of heat and electricity are properties which describe a(n)_______________ crystal.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What types of intermolecular forces exist between hydrogen fluoride molecules? I.London forces II) dipole-dipole interactions III) hydrogen bonding IV) ion-dipole interactions
A) II and III
B) I,II,and III
C) I only
D) III only
E) I and III
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
___________ ____________ is another name given for dispersion forces.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pure substances has the highest boiling point?
A) CF4
B) CCl4
C) CBr4
D) CI4
E) CH4
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms does not participate in hydrogen bonding?
A) S
B) O
C) F
D) N
E) H
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Ethanol (C2H5-OH)will have a greater viscosity than ethylene glycol (HO-CH2CH2-OH)at the same temperature.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Solids are generally most stable in crystalline form.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Which liquid is expected to have the larger surface tension at a given temperature,CCl4 or H2O? Briefly explain.
Correct Answer

verified
Water would have the...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 21 - 40 of 135
Related Exams